For reasons that have remained unclear, obese people are at a much higher risk for developing cancer compared to their thinner friends. The National Cancer Institute (NCI) estimates that around 85,000 new cancer cases each year are a direct result of obesity. While not yet up to the 171,000 or so new cases of lung cancer caused by smoking, obesity-related cancers are catching up. And as the number of smokers go down and the number of obese people go up, there may come a time in the future when obesity-related cancers are the most preventable ones.
Ideally, we would want to prevent these cancers by having obese people lose weight (but we’ve seen how successful this has been so far.)
Another way to reduce these numbers might be to figure out how obesity causes cancer. If we can do that, then maybe we can block the process and keep these cancers from forming in the first place. People can have their cake and eat it too!
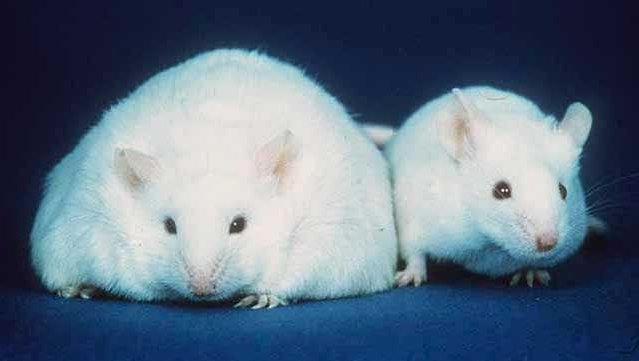
A new study out in Nature is a first step on this path (at least in mice). In this study, Yoshimoto and coworkers show that obesity by itself does not cause cancer in mice. Instead, bacteria specific to the gut of obese mice make certain cells more susceptible to getting cancer when exposed to carcinogens. Obesity makes the mice less able to deal with environmental insults.
The researchers first showed that in a protected environment, mice fed a high-fat diet were not at a higher risk for cancer compared to mice fed a low-fat diet. Both had about the same rates of cancer. So obesity itself does not directly cause cancer in these mice.
The situation is very different if these two groups of mice are exposed to a carcinogen (DMBA) while still in the womb. In that case, only around 5% of the mice fed the low-fat diet ended up with cancer while 100% of the mice fed the high-fat diet did. And the two groups ended up with different cancers—the thinner mice got lung cancer while the obese ones got liver cancer. Clearly something about obesity was making the livers of obese mice much more susceptible to the effects of the carcinogen.