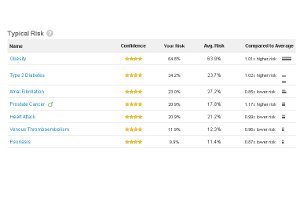
 Results like these get a company noticed by the FDA. (Click the image for a larger version.)
Results like these get a company noticed by the FDA. (Click the image for a larger version.)
A month or so ago a small company in San Diego struck a deal with Walgreens. Together they were going to sell genetic tests directly to consumers from drug store shelves. Sort of like how some paternity tests are now sold.
The resulting furor has caused Walgreens to back off. It has also awakened the slumbering beast—the FDA.
Last week the FDA sent a letter out to five different direct to consumer genetic testing companies about the genetic tests they offer. The FDA is arguing that these genetic tests are medical devices and therefore it has the authority to regulate them.
These tests actually consist of thousands of individual tests. If the FDA wants to regulate each one separately, there is no way that these companies will be able to make a profit. They’ll have to raise prices beyond what most people are willing to pay or simply fold up shop.
